TOPAMAX DANGERS
In March 2011, the Food and Drug Administration (FDA) released a warning regarding the increased risk of birth defects for pregnant women who take Topamax (topiramate). According to information reviewed by the North American Antiepileptic Drug Pregnancy Registry, Topamax increases the risk of oral clefts (cleft lip or cleft palate) nearly 4 times more than other antiepileptic drugs. The same study showed that Topamax users are 20 times more likely to give birth to children with oral clefts than women not taking an antiepileptic drug.
The FDA also moved Topamax to Pregnancy Category D, which means research indicates there is a possibility of harm to the fetus for humans. Topamax should only be prescribed when the benefit to the patient outweighs the risk, such as when not taking the drug would result in permanent injury or death.
TOPAMAX INJURIES
 Topamax is nearly 4 times more likely to cause cleft lip or cleft palate (oral clefts) in the children of women who take it while pregnant. These birth defects take place in the first trimester of pregnancy, often before the woman knows she is pregnant. This makes Topamax especially dangerous. The FDA recommends that women who are of childbearing age should use a birth control if they plan to take Topamax.
Topamax is nearly 4 times more likely to cause cleft lip or cleft palate (oral clefts) in the children of women who take it while pregnant. These birth defects take place in the first trimester of pregnancy, often before the woman knows she is pregnant. This makes Topamax especially dangerous. The FDA recommends that women who are of childbearing age should use a birth control if they plan to take Topamax.
Cleft lip and cleft palate, which can occur together, are caused when the lips or palate don’t fuse together properly, leaving an opening or a gap in the face. Oral cleft is a result of abnormal development in the first trimester, sometimes due to dangerous drugs.
FIXING A CLEFT LIP
At least one surgery is required to repair cleft lip. The surgery is expensive, and it often leaves scarring on the child’s face. Occasionally, more than one surgery is necessary to complete the reconstruction.
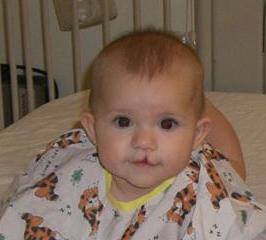 Damage caused by cleft palate is more extensive. Cleft palate can be corrected by surgery, but 3 out of 4 babies will need multiple surgeries. Often, as the child grows, more procedures will be necessary. Children with cleft palate will have difficulty talking and hearing, resulting in years of speech therapy. Other physical and psychological therapy is often needed to help children with oral clefts combat the effects of the condition.
Damage caused by cleft palate is more extensive. Cleft palate can be corrected by surgery, but 3 out of 4 babies will need multiple surgeries. Often, as the child grows, more procedures will be necessary. Children with cleft palate will have difficulty talking and hearing, resulting in years of speech therapy. Other physical and psychological therapy is often needed to help children with oral clefts combat the effects of the condition.
HISTORY OF TOPAMAX
Topamax (topiramate) is an anticonvulsant drug that is also approved by the FDA to prevent migraines. It was first marketed in 1996 by subsidiaries of Johnson & Johnson for its treatment of certain types of seizures. In 2010, Johnson & Johnson lost a lawsuit, which required them to pay $81 million for illegally promoting Topamax for psychiatric uses, including the treatment of bipolar disorder. Topamax is not FDA-approved for any psychiatric uses.
Topiramate was shown early on to have teratogenic (defects in the development) effects in the offspring of animals. In 2008, the first study was conducted linking Topamax to a risk of oral cleft birth defects. The study used only a small group of epilepsy patients, but it served to stimulate further research.
Three years later, in March of 2011, the FDA released its warning based on a study conduced by the North American Antiepileptic Drug Pregnancy Registry. According to the FDA, 4.3 million patients filled topiramate prescriptions in the U.S. from 2007 to 2010 alone. Hopefully future birth defects can be avoided with the FDA warning and with Topamax being placed in Pregnancy Category D. However, Topamax has been on the market for fifteen years, and families have already suffered the devastating effects of this dangerous drug. If you took Topamax and your baby was born with an oral cleft, you deserve compensation.
Have you or a loved one suffered Topamax side effects?
At The Oshman Firm, our experienced Topamax attorneys will provide you with aggressive representation in the handling of your case. If your infant suffered oral cleft birth defects that may have been caused by Topamax, call us at 800-400-8182 or contact us via email to represent your interests in a Topamax lawsuit. We offer a free, no-obligation consultation with a Topamax lawyer so you can learn more about your legal rights and options.
